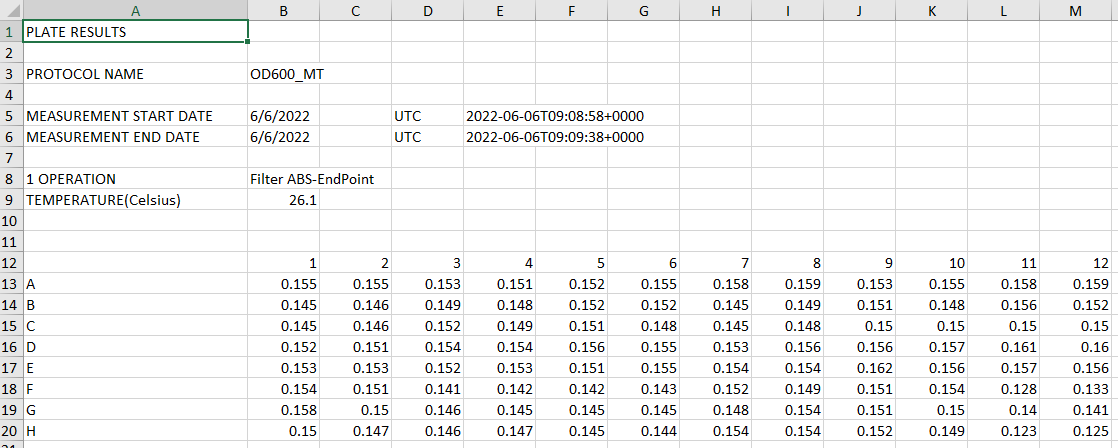
- EuPathDB: Provides access to pathogen annotation resources available on EuPathDB databases
This package helps us to retrieve annotation data from EuPathDB.
- AnnotationForge: Tools for building SQLite-based annotation data packages
Databases for fungal pathogens were provided on different platforms, we need a way to make it compatible to input into other analysis packages. Annotation Forge is a robust tool for this task.
- AnnotationHub: Client to access AnnotationHub resources
The Bioconductor team wants to collect popular web resources into their package called AnnotationHub. This beneficial tool provides genomic and transcriptomic profiles for many applications.
- Biomart: Interface to BioMart databases (i.e. Ensembl)
This tool provides a way to access BioMart (Ensembl) annotation database directly from R. This is very useful to query annotation data from Ensembl.
- clusterProfiler: A universal enrichment tool for interpreting omics data
This package provides an interface for functional annotation using data from various sources. They also provide excellent functions to visualize enrichment results obtained from its analysis.
- hpgltools: A pile of (hopefully) useful R functions
This is a set of functions really helpful for host-pathogen interaction analysis.
One of my very first bioinformatic tutors develop it!!! I should say that the author of this package is a super lovely scientist. I was an amateur guy in bioinformatics when I started my Ph.D. There were so many simple troubles I was struggling with every day. I have some problems with the database on AnnotationHub. I contacted him, and he was so helpful and kind to help me a lot. He made an Rmd file to guide me in handling my data and try to explain basic things via email. I am very grateful to him. Hope to see him in person somewhere soon!